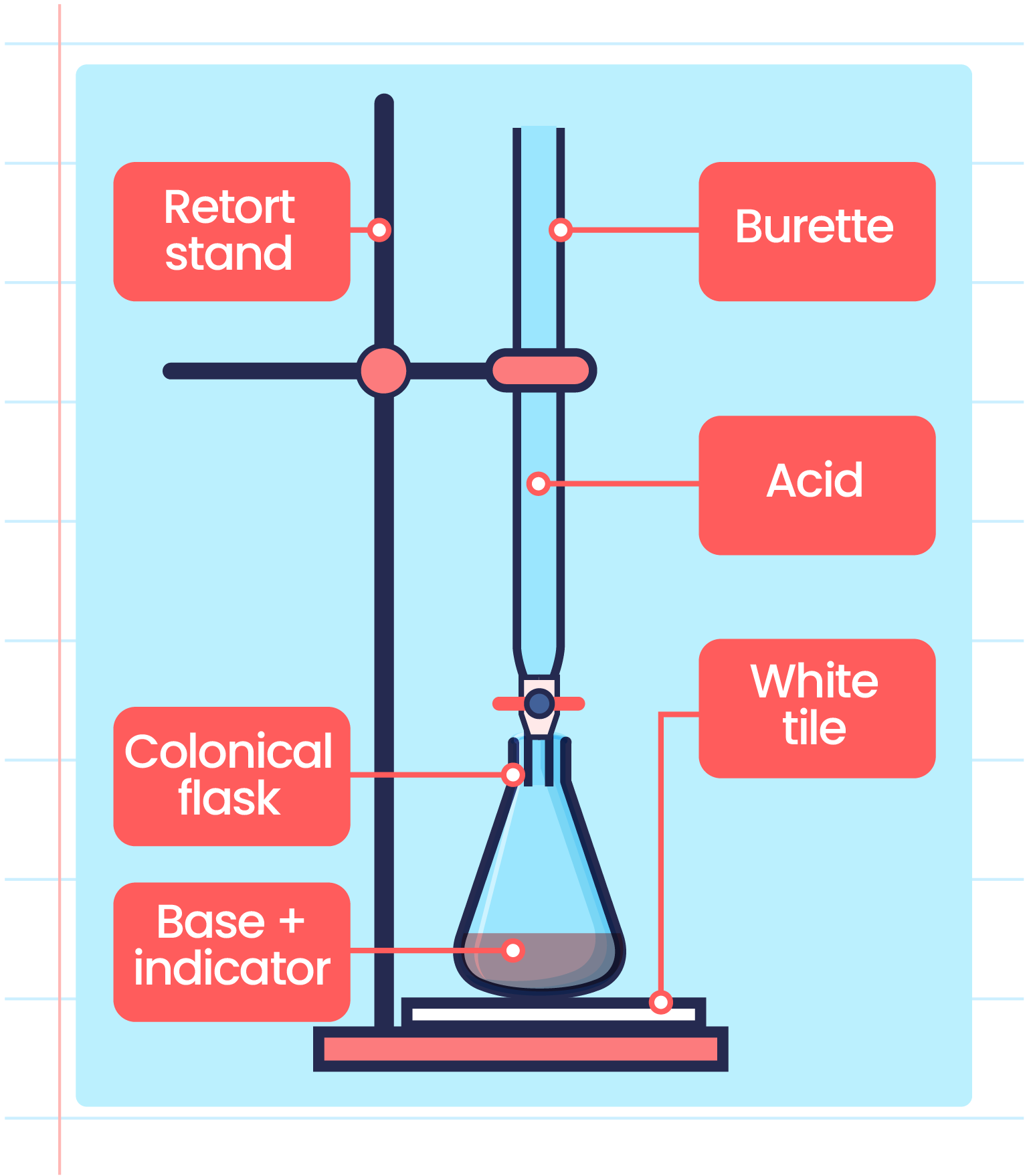
Acid Base Titration Neutralization Reaction .the solution is neutral, having a ph = 7.00.one point in the titration of a weak acid or a weak base is particularly important:
from fity.club
This involves the addition of titrant in excess of the.one point in the titration of a weak acid or a weak base is particularly important:the solution is neutral, having a ph = 7.00.
Neutralisation Meaning
Acid Base Titration Neutralization Reaction Before we start discussing about titration and titration. Before we start discussing about titration and titration. (d) titrant volume = 37.50 ml.one point in the titration of a weak acid or a weak base is particularly important:
From www.youtube.com
Acids and Bases, Titrations, and Neutralization Reactions YouTube Acid Base Titration Neutralization Reaction Before we start discussing about titration and titration.one point in the titration of a weak acid or a weak base is particularly important: The midpoint of a titration is defined as the. This involves the addition of titrant in excess of the. (d) titrant volume = 37.50 ml. Acid Base Titration Neutralization Reaction.
From www.scienceabc.com
Titration Chemistry Definition, Explanation, Formula And Calculation Acid Base Titration Neutralization Reactionthe solution is neutral, having a ph = 7.00. (d) titrant volume = 37.50 ml. In a titration, a solution of known concentration (the titrant) is added to a solution of the.one point in the titration of a weak acid or a weak base is particularly important: Before we start discussing about titration and titration. Acid Base Titration Neutralization Reaction.
From www.slideserve.com
PPT Neutralization Reactions PowerPoint Presentation, free download Acid Base Titration Neutralization Reactionone point in the titration of a weak acid or a weak base is particularly important:the solution is neutral, having a ph = 7.00. The midpoint of a titration is defined as the. In a titration, a solution of known concentration (the titrant) is added to a solution of the. This involves the addition of titrant in. Acid Base Titration Neutralization Reaction.
From www.slideserve.com
PPT AcidBase Titrations A. Definition volumetric analysis of the Acid Base Titration Neutralization Reaction In a titration, a solution of known concentration (the titrant) is added to a solution of the. The midpoint of a titration is defined as the. Before we start discussing about titration and titration. (d) titrant volume = 37.50 ml.the solution is neutral, having a ph = 7.00. Acid Base Titration Neutralization Reaction.
From sachiacidbase.weebly.com
Titrations Sachi's Acids and Bases Acid Base Titration Neutralization Reaction Before we start discussing about titration and titration. In a titration, a solution of known concentration (the titrant) is added to a solution of the.the solution is neutral, having a ph = 7.00. This involves the addition of titrant in excess of the.one point in the titration of a weak acid or a weak base is. Acid Base Titration Neutralization Reaction.
From www.slideserve.com
PPT AcidBase Titration & pH Chapter 15 PowerPoint Presentation ID Acid Base Titration Neutralization Reaction (d) titrant volume = 37.50 ml. In a titration, a solution of known concentration (the titrant) is added to a solution of the. Before we start discussing about titration and titration.the solution is neutral, having a ph = 7.00.one point in the titration of a weak acid or a weak base is particularly important: Acid Base Titration Neutralization Reaction.
From general.chemistrysteps.com
Titration of a Weak Acid by a Strong Base Chemistry Steps Acid Base Titration Neutralization Reaction (d) titrant volume = 37.50 ml. In a titration, a solution of known concentration (the titrant) is added to a solution of the. Before we start discussing about titration and titration. The midpoint of a titration is defined as the. This involves the addition of titrant in excess of the. Acid Base Titration Neutralization Reaction.
From www.chemicals.co.uk
A Level Chemistry Revision Physical Chemistry Acids And Bases Acid Base Titration Neutralization Reaction The midpoint of a titration is defined as the. (d) titrant volume = 37.50 ml. In a titration, a solution of known concentration (the titrant) is added to a solution of the. Before we start discussing about titration and titration.the solution is neutral, having a ph = 7.00. Acid Base Titration Neutralization Reaction.
From pressbooks.online.ucf.edu
15.7 AcidBase Titrations Chemistry Fundamentals Acid Base Titration Neutralization Reaction This involves the addition of titrant in excess of the. Before we start discussing about titration and titration. In a titration, a solution of known concentration (the titrant) is added to a solution of the. (d) titrant volume = 37.50 ml.the solution is neutral, having a ph = 7.00. Acid Base Titration Neutralization Reaction.
From present5.com
AcidBase Titrations Barb Fallon AP Chemistry June 2007 Acid Base Titration Neutralization Reaction Before we start discussing about titration and titration.one point in the titration of a weak acid or a weak base is particularly important: The midpoint of a titration is defined as the. This involves the addition of titrant in excess of the. In a titration, a solution of known concentration (the titrant) is added to a solution of. Acid Base Titration Neutralization Reaction.
From www.slideserve.com
PPT Neutralization Reactions using Titration Method PowerPoint Acid Base Titration Neutralization Reactionone point in the titration of a weak acid or a weak base is particularly important:the solution is neutral, having a ph = 7.00. The midpoint of a titration is defined as the. Before we start discussing about titration and titration. (d) titrant volume = 37.50 ml. Acid Base Titration Neutralization Reaction.
From webmis.highland.cc.il.us
AcidBase Titrations Acid Base Titration Neutralization Reactionthe solution is neutral, having a ph = 7.00. In a titration, a solution of known concentration (the titrant) is added to a solution of the.one point in the titration of a weak acid or a weak base is particularly important: (d) titrant volume = 37.50 ml. The midpoint of a titration is defined as the. Acid Base Titration Neutralization Reaction.
From psu.pb.unizin.org
14.7 AcidBase Titrations Chemistry 112 Chapters 1217 of OpenStax Acid Base Titration Neutralization Reactionone point in the titration of a weak acid or a weak base is particularly important:the solution is neutral, having a ph = 7.00. The midpoint of a titration is defined as the. This involves the addition of titrant in excess of the. In a titration, a solution of known concentration (the titrant) is added to a. Acid Base Titration Neutralization Reaction.
From cetyps.pics
Titration of a Weak Base by a Strong Acid Chemical Steps (2022) Acid Base Titration Neutralization Reaction The midpoint of a titration is defined as the. Before we start discussing about titration and titration. This involves the addition of titrant in excess of the. (d) titrant volume = 37.50 ml. In a titration, a solution of known concentration (the titrant) is added to a solution of the. Acid Base Titration Neutralization Reaction.
From www.chegg.com
Solved Using the following pH curve for the titration of a Acid Base Titration Neutralization Reaction This involves the addition of titrant in excess of the.the solution is neutral, having a ph = 7.00. Before we start discussing about titration and titration. (d) titrant volume = 37.50 ml.one point in the titration of a weak acid or a weak base is particularly important: Acid Base Titration Neutralization Reaction.
From study.com
Neutralization Reaction Definition, Equation & Examples Lesson Acid Base Titration Neutralization Reaction (d) titrant volume = 37.50 ml. This involves the addition of titrant in excess of the. Before we start discussing about titration and titration. The midpoint of a titration is defined as the.one point in the titration of a weak acid or a weak base is particularly important: Acid Base Titration Neutralization Reaction.
From www.tutormyself.com
233 (Triple only) describe how to carry out an acidalkali titration Acid Base Titration Neutralization Reaction The midpoint of a titration is defined as the.one point in the titration of a weak acid or a weak base is particularly important: Before we start discussing about titration and titration. This involves the addition of titrant in excess of the. (d) titrant volume = 37.50 ml. Acid Base Titration Neutralization Reaction.
From chem.libretexts.org
17.4 Neutralization Reactions and Titration Curves Chemistry LibreTexts Acid Base Titration Neutralization Reaction (d) titrant volume = 37.50 ml.one point in the titration of a weak acid or a weak base is particularly important: This involves the addition of titrant in excess of the. Before we start discussing about titration and titration. The midpoint of a titration is defined as the. Acid Base Titration Neutralization Reaction.